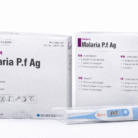
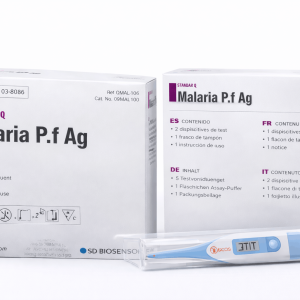
MALARIA TEST KITS
Malaria Test Kit (Malaria P.f Ag) is a rapid diagnostic test designed for the qualitative detection of Plasmodium falciparum antigen in human blood. It provides fast, reliable results and is suitable for clinical and field use.
- Estimated Delivery : Up to 4 business days
- Free Shipping & Returns : On all orders over $200
Malaria Test Kit – Malaria P.f Ag is a rapid immunochromatographic diagnostic test intended for the qualitative detection of Plasmodium falciparum (P.f) antigen in whole blood samples. The test is designed to aid in the early diagnosis of malaria infections, allowing for timely treatment and effective disease management.
The test kit delivers quick results within minutes and does not require complex laboratory equipment, making it ideal for use in hospitals, clinics, laboratories, and remote or field settings. Its simple testing procedure ensures ease of use by trained healthcare professionals.
Each kit typically contains 25 test devices, assay diluent, and detailed instructions for use. The product is manufactured under strict quality standards and is intended for in-vitro diagnostic use only.
✅ Key Features:
-
Rapid Diagnostic Test for Malaria
-
Detects Plasmodium falciparum (P.f) Antigen
-
Fast & Accurate Results
-
Easy to Use
-
Suitable for Clinical, Laboratory & Field Use
-
In-Vitro Diagnostic (IVD) Use Only
-
Reliable & High-Quality Performance
Malaria Test Kit (P.f Ag) is an essential diagnostic tool for healthcare facilities seeking a quick, dependable, and cost-effective solution for malaria screening and diagnosis.










Reviews
There are no reviews yet.